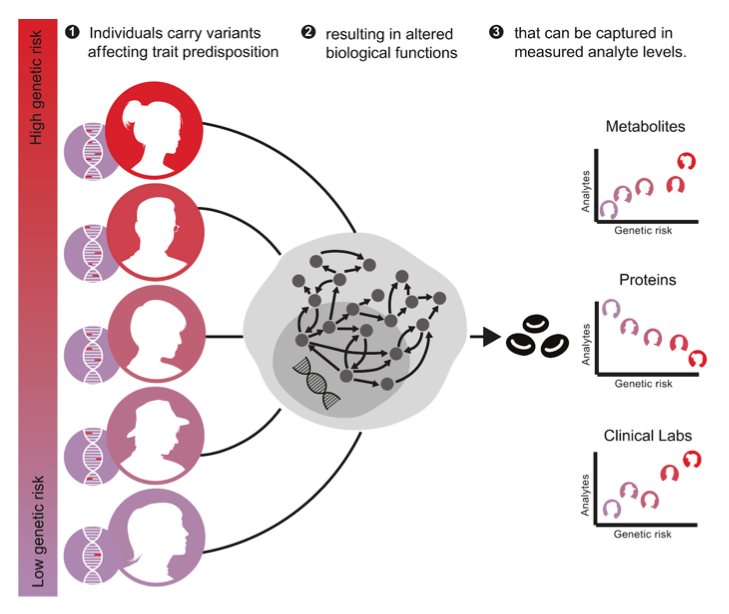
Transitions from health to disease are characterized by dysregulation of biological networks under the influence of genetic and environmental factors, often over the course of years to decades before clinical symptoms appear. Understanding these dynamics has important implications for preventive medicine. However, progress has been hindered both by the difficulty of identifying individuals who will eventually go on to develop a particular disease, and by the inaccessibility of most disease-relevant tissues in living individuals. We have developed an alternative approach using polygenic risk scores (PRSs) based on genome-wide association studies (GWAS), initially for 54 diseases and complex traits coupled with multi-omic profiling, and find that genetic risk for these traits is associated with hundreds of detectable alterations in plasma proteomic, metabolomic and clinical laboratory measurements across several thousand mostly healthy individuals. We recapitulate a variety of known relationships (e.g. depression with glutaminergic neurotransmission and inflammation, IL-33 with asthma risk) and find novel associations directly suggesting therapeutic strategies and protection from aging . Our results provide insights into the molecular pathophysiology of a wide range of traits and demonstrate how using genetic risk as a proxy for prodromal disease can substantially broaden the insights gleaned from population-scale multi-omic cohorts.
Since every individual has a calculable PRS for every disease (with quality GWAS data), every individual can inform the manifestation of every disease, dramatically broadening inclusion and deepening insight.
Current Project Leads:
 |  |  |
| Michael Wainberg | Nathan Price | Leroy Hood |



 hood-price.isbscience.org/research/manifestation-of-polygenic-risk-scores/
hood-price.isbscience.org/research/manifestation-of-polygenic-risk-scores/