Alheimer’s Disease and Brain Health is an active area of research in our lab. Alzheimer’ Disease is the 6th leading cause of death in the US, with a high financial and health burden on sufferers and family caregivers and, astonishingly, over 400 drug trials have essentially failed to treat the disease. We focus on the molecular neurochemistry of Alzheimer’s Disease with funding from the NIA through the AMP-AD consortium (Accelerating Medicines Partnership – Alzheimer’s Disease) as well as muti-modal Clinical Trials for the treatment of Alzheimer’s Disease through our partnerships with the Pacific Neuroscience Institute and the Hoag within the Providence St. Joseph Health System as well as the Brain Health Research Institute in Seattle.
We are identifying molecular networks underlying Alzheimer’s Disease, in order to understand disease etiology and discover novel therapeutic targets through collaboration with academic and pharma partners in AMP-AD
A Systems Approach to Targeting Innate Immunity in Alzheimer’s Disease seeks to validate therapeutic targets, identify molecular mechanisms that mediate disease transition to Pathological AD, and share our data openly with the larger scientific community in order to provide biologic insight into the mechanism of action of the proposed targets and inform on the direction of change needed for therapeutic benefit. PI Todd Golde, University of Florida; Co-I Nathan Price, ISB; Co-I Nilufer Ertekin-Taner, Mayo Clinic. Project period, 2018-2023.
Open Systems for AMP-AD Target Enhancement seeks to develop a visual dashboard and integrative analysis platform in support of cross-consortium AMP- AD target prioritization; develop temporal models of disease progression; and develop in silico high throughput drug screening tools to support network-based target prioritization and drug discovery efforts. The proposed work is designed to maximize the vast resources and biological insights within AMP-AD and to accelerate characterization and validation of AMP-AD targets through the development and open dissemination of a set of consortium-wide high-confidence therapeutic targets in conjunction with the computational and experimental tools necessary for their validation. PI Lara Mangravite, Sage Bionetworks; Co-I Nathan Price, ISB; Co-I Paul Brennan, Sage Bionetworks; Co-I Eric Schadt, Sage Bionetworks. Project Period, 2017-2020.
Metabolic Network Analysis of Biochemical Trajectories in Alzheimer’s Disease seeks to profile longitudinal samples from a cohort where deep phenotype data exists in order to track early biochemical changes related to both CSF amyloid-beta and tau pathology across the trajectory of disease; create an integrated molecular Atlas for AD; build the first genome-scale model of metabolic changes underlying AD progression. Together, this effort will provide a biochemical roadmap towards effective early interventions for AD. PI Rima Kaddurah-Daouk, Duke University; Co-I Nathan Price, ISB; Co-I Gabi Kastenmuller, Duke University. Project Period, 2017-2020.
Metabolic Signatures for Disease Sub-Classification and Target Prioritization in Alzheimer’s Disease seeks to expand our previous metabolomic analyses to accelerate AMP-AD progress towards novel drug discovery, systematically addressing contributions of peripheral metabolism to brain health and disease and provide biochemical readouts as an intermediate phenotype for rich omics data generated in the consortium. This will provide an enabling metabolic interconnecting framework to accelerated AD therapeutic developing in AMP-AD. PI Rima Kaddurah-Daouk, Duke University; Co-I Nathan Price, ISB; Co-I Gabe Kastenmuller, Duke University; Co-I Cornelia Marja Van Duijn. Project Period 2018-2023.
Other research areas important for Brain Health include on-going Department of Defense funded studies to identify biomarkers of PTSD predictive of the trajectory of the disorder; and an industry funded project leveraging PD3 clouds to understand Multiple Sclerosis.
Current Clinical trials in Alzheimer’s Disease include:
Coaching for Cognition in Alheimer’s Disease (COCOA) is a multimodal therapeutic trial evaluating wellness coaching and brain training with standard care relative to standard care alone.
Precision Recommendations to Optimize Neurotransmission (PREVENTION) is a trial of data-driven multimodal lifestyle interventions for the treatment of Alzheimer’s Disease
Selected recent publications describing this work include:
Multi-omic biomarker identification and validation for diagnosing warzone-related post-traumatic stress disorder. Dean KR, Hammamieh R, Mellon SH, Abu-Amara D, Flory JD, Guffanti G, Wang K, Daigle BJ Jr, Gautam A, Lee I, Yang R, Almli LM, Bersani FS, Chakraborty N, Donohue D, Kerley K, Kim TK, Laska E, Young Lee M, Lindqvist D, Lori A, Lu L, Misganaw B, Muhie S, Newman J, Price ND, Qin S, Reus VI, Siegel C, Somvanshi PR, Thakur GS, Zhou Y; PTSD Systems Biology Consortium, Hood L, Ressler KJ, Wolkowitz OM, Yehuda R, Jett M, Doyle FJ 3rd, Marmar C. Mol Psychiatry. 2019 Sep 10. doi: 10.1038/s41380-019-0496-z. PMID: 31501510
Distinct Profiles of Cell-Free MicroRNAs in Plasma of Veterans with Post-Traumatic Stress Disorder. Lee MY, Baxter D, Scherler K, Kim TK, Wu X, Abu-Amara D, Flory J, Yehuda R, Marmar C, Jett M, Lee I, Wang K, Hood L. J Clin Med. 2019 Jul 3;8(7). pii: E963. doi: 10.3390/jcm8070963. PMID: 31277223
Multiscale Analysis of Independent Alzheimer’s Cohorts Finds Disruption of Molecular, Genetic, and Clinical Networks by Human Herpesvirus. Readhead B, Haure-Mirande JV, Funk CC, Richards MA, Shannon P, Haroutunian V, Sano M, Liang WS, Beckmann ND, Price ND, Reiman EM, Schadt EE, Ehrlich ME, Gandy S, Dudley JT. Neuron. 2018 Jul 11;99(1):64-82.e7. doi: 10.1016/j.neuron.2018.05.023. Epub 2018 Jun 21. PMID: 29937276
Conserved brain myelination networks are altered in Alzheimer’s and other neurodegenerative diseases. Allen M, Wang X, Burgess JD, Watzlawik J, Serie DJ, Younkin CS, Nguyen T, Malphrus KG, Lincoln S, Carrasquillo MM, Ho C, Chakrabarty P, Strickland S, Murray ME, Swarup V, Geschwind DH, Seyfried NT, Dammer EB, Lah JJ, Levey AI, Golde TE, Funk C, Li H, Price ND, Petersen RC, Graff-Radford NR, Younkin SG, Dickson DW, Crook JR, Asmann YW, Ertekin-Taner N. Alzheimers Dement. 2018 Mar;14(3):352-366. Doi: 10.1016/j.jalz.2017.09.012. Epub 2017 Oct 31. PMID: 29107053
Predictive Big Data Analytics: A Study of Parkinson’s Disease Using Large, Complex, Heterogeneous, Incongruent, Multi-Source and Incomplete Observations. Dinov ID, Heavner B, Tang M, Glusman G, Chard K, Darcy M, Madduri R, Pa J, Spino C, Kesselman C, Foster I, Deutsch EW, Price ND, Van Horn JD, Ames J, Clark K, Hood L, Hampstead BM, Dauer W, Toga AW. PLoS One. 2016 Aug 5;11(8):e0157077. doi: 10.1371/journal.pone.0157077. eCollection 2016. PMID: 27494614
Big biomedical data as the key resource for discovery science. Toga AW, Foster I, Kesselman C, Madduri R, Chard K, Deutsch EW, Price ND, Glusman G, Heavner BD, Dinov ID, Ames J, Van Horn J, Kramer R, Hood L. J Am Med Inform Assoc. 2015 Nov;22(6):1126-31. doi: 10.1093/jamia/ocv077. Epub 2015 Jul 21. PMID: 26198305
IL-10 alters immunoproteostasis in APP mice, increasing plaque burden and worsening cognitive behavior. Chakrabarty P, Li A, Ceballos-Diaz C, Eddy JA, Funk CC, Moore B, DiNunno N, Rosario AM, Cruz PE, Verbeeck C, Sacino A, Nix S, Janus C, Price ND, Das P, Golde TE. Neuron. 2015 Feb 4;85(3):519-33. doi: 10.1016/j.neuron.2014.11.020. Epub 2015 Jan 22. PMID: 25619653
Featured Projects
-

Exploring the Potential of Viral Contributors to Alzheimer’s Disease
Alzheimer ‘s disease (AD) affects millions of Americans and causes significant morbidity and mortality. Although genetic determinants of AD have been a major focus of research over the last three decades, there is limited insight into co-factors that contribute to AD pathology and progression. Recent genetic associations implicating alterations in innate immunity to risk for AD, suggest that environmental factors, such as infection, may modulate brain immune function and could…
-

Biomarkers for Post-Traumatic Stress Disorder
Blood based diagnostic marker for Post-traumatic stress disorder (PTSD) Post-traumatic stress disorder (PTSD) poses a significant burden on emotional and physical health and health care costs. According to US Department of Veterans Affairs, about 12% of Gulf war veterans, including those deployed to Iraq and Afghanistan, suffer from PTSD in a given year and about 30% of Vietnam veterans have had PTSD in their lifetime. The annual treatment cost for…
-

PHEWAS Approach to Study Alzheimer’s Disease
Genetics play an important role in late-onset Alzheimer’s Disease (AD) etiology. As AD develops over decades, elucidating the biological effects of AD-associated genetic variation across the adult lifespan may illuminate underlying processes in the causal pathway for AD, and potentially provide novel mid-life targets for behavioral and drug intervention. To address this, we are utilizing high dimensional, high-quality measurements from thousands of participants in a wellness program, aged 18 to…
-
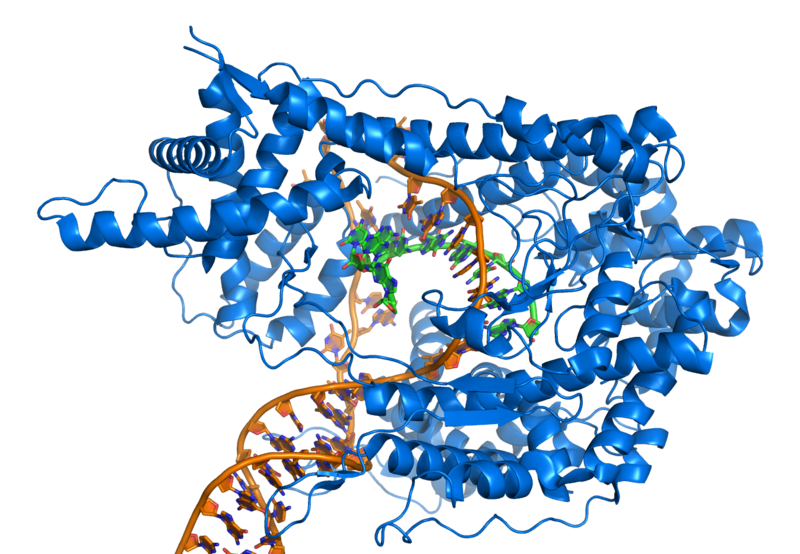
Transcriptional Regulatory Networks in Alzheimer’s Disease
We are using Transcriptional Regulatory Networks (TRNs) to better understand the causes and progression of Alzheimer’s disease. Our approach starts with measurements of gene expression in the brain, from which we create models that help to understand the changes in transcriptional regulatory structure that occur in Alzheimer’s disease. We are developing methods that help to identify cell-type specific changes in gene expression, as well as changes in gene regulation by transcription factors….



 hood-price.isbscience.org/research/alzheimers-disease-and-brain-health/
hood-price.isbscience.org/research/alzheimers-disease-and-brain-health/